Hot Issues
Take action!
Contact your legislators to stop fake online pharmacies and report social media drug sellers to the DEA
Follow us: Twitter | YouTube | Facebook | LinkedIn | Instagram
This is a reprint of an FDA Alert. STOP CLOPEZ CORP Issues Voluntary Worldwide Recall of Schwinnng Capsules Due to the Presence of Undeclared Nortadalafil When a company announces a recall, market withdrawal, or safety alert, the FDA posts the company’s announcement as a public service. FDA does not endorse either the product or the…
The Southern District of New York sentenced criminals from two separate HIV drug diversion rings.
The products appear to have been purchased from unlicensed sources. Medications purchased from unlicensed sources may be misbranded, adulterated, counterfeit, contaminated, improperly stored and transported, ineffective and/or unsafe.
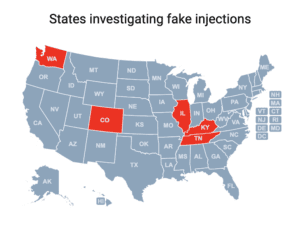
Cases of a botulism-like illness linked to cosmetic injections have been reported in Colorado, Illinois, Kentucky, Tennessee and Washington.
The state of Florida provided these FOIA-responsive documents to PSM for the state’s final and ultimately approved application to establish a Canadian drug importation program.
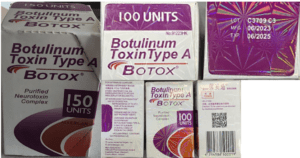
People in nine states sought medical treatment and some were hospitalized after receiving injections of fake Botox in what the CDC has characterized as “non-medical settings.”
Our podcast covers the latest in pharma crime and medicine safety.
Like your information on video? Subscribe to our YouTube playlist!

A gang in India sold fake cancer treatments in India, China and the U.S. Watch.

A Maryland company settled in Gilead Sciences' lawsuit over counterfeit HIV drugs.

A supplement company must pay $2.4 million for selling unapproved drugs, including tianeptine, which is banned in 9 states.
Click the images below to see more recent videos.